CARBOHYDRATE STRUCTURE GENERALIZATION SCHEME FOR DATABASE-DRIVEN SIMULATION OF EXPERIMENTAL OBSERVABLES, SUCH AS NMR CHEMICAL SHIFTS
aHigher Chemical College of the Russian Academy of Sciences, Moscow, Russia
bN.D. Zelinsky Institute of Organic Chemistry, Russian Academy of Sciences, Moscow, Russia
KEYWORDS: NMR simulation, 13C NMR, carbohydrate, saccharide, statistical approach, database, glycan, chemical shift
Journal of Chemical Information and Modeling, 2014, v. 54(9), pp.2594–2611
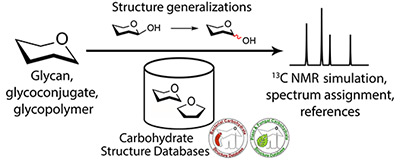 Carbohydrates play an immense role in different aspects of life. NMR spectroscopy is a major and the most powerful tool for investigation of these compounds. Nowadays, progress in computational procedures has opened up novel opportunities, which gave an impulse to develop new instruments intended to make the research simpler and more efficient. In this paper, we present a new approach to simulate 13C NMR chemical shifts of carbohydrates. The approach is suitable for any atomic observables, which could be stored in a database. The method is based on sequential generalization of the chemical surrounding of the atom under prediction and heuristic averaging of database data. Unlike existing applications, the generalization schema is tuned for carbohydrates, including those containing phosphates, amino acids, alditols, and other non-carbohydrate constituents. It was implemented in the GODESS (Glycan-Optimized Dual Empirical Spectrum Simulation) software freely available on the Internet. In the field of carbohydrates, our approach was shown to outperform all other existing methods of NMR spectrum prediction (including quantum mechanical calculations) in accuracy. Only this approach supports NMR spectrum simulation for a number of structural features in polymeric structures.
Carbohydrates play an immense role in different aspects of life. NMR spectroscopy is a major and the most powerful tool for investigation of these compounds. Nowadays, progress in computational procedures has opened up novel opportunities, which gave an impulse to develop new instruments intended to make the research simpler and more efficient. In this paper, we present a new approach to simulate 13C NMR chemical shifts of carbohydrates. The approach is suitable for any atomic observables, which could be stored in a database. The method is based on sequential generalization of the chemical surrounding of the atom under prediction and heuristic averaging of database data. Unlike existing applications, the generalization schema is tuned for carbohydrates, including those containing phosphates, amino acids, alditols, and other non-carbohydrate constituents. It was implemented in the GODESS (Glycan-Optimized Dual Empirical Spectrum Simulation) software freely available on the Internet. In the field of carbohydrates, our approach was shown to outperform all other existing methods of NMR spectrum prediction (including quantum mechanical calculations) in accuracy. Only this approach supports NMR spectrum simulation for a number of structural features in polymeric structures.